Research Overview
Our laboratory studies the sympathetic nervous system, which is a key regulator of whole body physiology in animals. The sympathetic nervous system is critical for triggering physiological responses to stress/danger and in maintaining body homeostasis under basal conditions. Sympathetic axons innervate diverse peripheral organs and tissues to control fundamental processes that affect our daily lives, including heart rate, body temperature, blood glucose levels, and immune function. We study sympathetic neurons because they serve as an excellent paradigm to understand how peripheral organs and tissues guide neuronal development, communication, and repair. We are also motivated by the growing awareness that sympathetic dysfunction underlies several pathologies including peripheral neuropathies, heart failure, hypertension, and diabetes. Our group has identified key molecular and cellular mechanisms by which peripheral organs instruct the development of sympathetic neurons through regulating neuron survival, axon growth, and target innervation. In recent work, we have addressed the contribution of sympathetic input to organ development and function by focusing on the pancreatic islets. Together, our studies of the sympathetic nervous system using mouse genetics, imaging, cellular, biochemical, and functional analyses provide new insights into the interplay between the nervous system and other organ systems.
Research Projects
a. Axonal transport of neurotrophin receptors regulates sympathetic neuron development and function
Ever since the pioneering studies of Rita Levi-Montalcini, Viktor Hamburger, and colleagues, neurobiologists have appreciated the importance of target tissues in neuronal development. Their work laid the foundation for a central tenet in developmental neuroscience, the ‘neurotrophic factor hypothesis’, that postulates that neurons are over-produced during development, and compete for limiting amounts of target-derived factors to grow, survive, and innervate target tissues. To date, the neurotrophin, nerve growth factor (NGF), is the best characterized target-derived instructive cue for sympathetic neuron development. As NGF is released by neuronal targets, a key cell biological problem is to understand how a signal impinging on axon terminals is relayed to trigger cellular responses necessary for neuronal development. We found that NGF signaling is communicated via vesicular transport of internalized NGF:TrkA receptor complexes in sympathetic neurons (Scott-Solomon et al., 2021). We have identified key mechanisms of NGF receptor endocytosis and axonal transport in sympathetic neurons. For example, we found that TrkA endocytosis in axons relies on the calcium-responsive phosphatase, calcineurin, and dephosphorylation of neuron-specific splicing isoforms of dynamin1 (Bodmer et al., 2011). This finding led us to identify a role for dysregulated TrkA trafficking in Down syndrome, caused by trisomy of human chromosome 21. One of the genes that is triplicated in Down syndrome is Regulator of Calcineurin1 (RCAN1), an endogenous inhibitor of calcineurin. In a mouse model of Down syndrome, we observed impaired TrkA trafficking, loss of sympathetic neurons, and reduced innervation of targets, all of which could be alleviated by genetically reducing RCAN1 levels (Patel et al., 2015).
Neuronal responsiveness to target-derived factors also requires the precise axonal targeting of new receptors, synthesized in cell bodies. We discovered that TrkA receptors are delivered to sympathetic axons by a non-canonical transport mechanism called transcytosis (Ascano et al., 2009; Yamashita et al., 2017). Transcytosis is an atypical endocytosis-based mechanism, where newly synthesized proteins are first inserted on soma surfaces, internalized, and recycled long-distance to axons. Remarkably, transcytosis of TrkA receptors is regulated by NGF itself acting on axons, suggesting a positive feedback mechanism to scale up receptor availability in axons at times of need.
Current studies: We are using live imaging, confocal and electron microcopy, and cell biological/biochemical analyses to monitor the dynamic behavior and transport kinetics of receptor transcytosis, uncover the identity of the organelles responsible for TrkA transcytosis, and underlying regulatory mechanisms. An attractive hypothesis is that transcytosis is a more general mechanism than currently appreciated for the axonal delivery of pre-synaptic membrane proteins. We are testing this hypothesis using a biotinylation-based proteomic screen.
Yamashita et al., 2017
b. Sympathetic innervation promotes pancreatic islet maturation
The past decade has seen an exponentially growing interest in the sympathetic nervous system from the broader perspective of understanding how neural innervation affects target organ development and function. We have chosen to address this question by focusing on pancreatic islets, the functional units responsible for maintaining blood glucose levels. Islets are richly innervated by sympathetic nerves, and sympathetic input is known to modulate islet hormone release and influence glucose homeostasis in adult life (Lin et al., 2021). However, the developmental role(s) of innervation is far less defined. We provided the first evidence that early loss of sympathetic nerves disrupts islet morphology in neonatal mice and results in reduced insulin secretion and impaired glucose tolerance in adults (Borden et al., 2013).
Current studies: We are investigating anatomical, cellular, and functional interactions between sympathetic nerves and pancreatic cells using high resolution imaging, genomic and biochemical approaches, and genetic mouse models. To gain an unbiased view of islet transcriptional changes induced by de-innervation, we are using single-cell RNA sequencing. Viral tracing and transcriptional profiling allow us to map the neural circuits that regulate islet function and characterize the molecular identity of pancreas-projecting sympathetic neurons. We are also exploring a role for nerve-derived Neuropeptide Y (NPY) in islet organization and functional maturation. Such studies will provide critical information about the impact of sympathetic input on islet health and, potentially, lead to neuromodulation-based strategies for treatment of diabetes.
Kumari et al., 2023
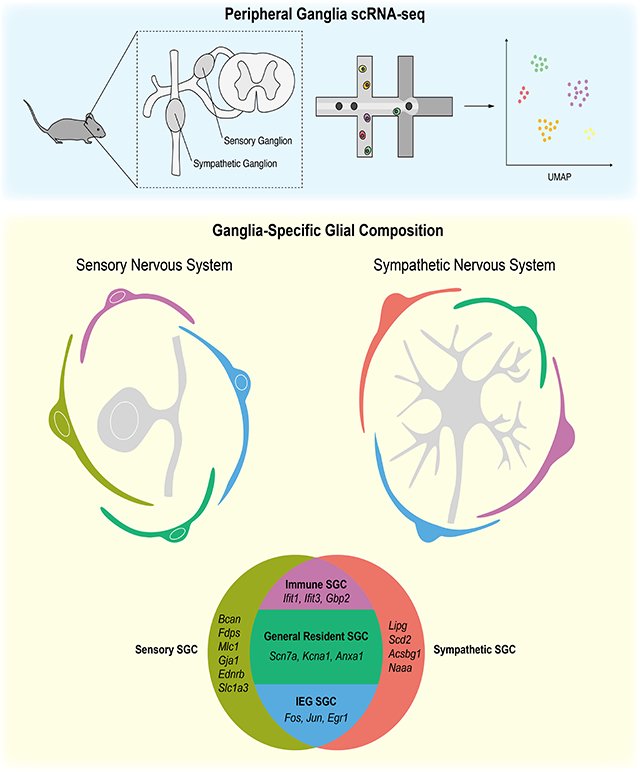
c. Satellite glial cells influence the activity of sympathetic neurons
Satellite glia are the major glial cell type present in the ganglia of the peripheral nervous system, where they envelop neuronal cell bodies. Despite this intimate association and emerging evidence implicating these cells in chronic pain and cardiac dysfunction, sateliite glial cells are one of the least understood cell types in the nervous system. Using single cell RNA sequencing and fluorescence in situ hybridization, we showed that satellite glia in sympathetic and sensory ganglia are remarkably diverse and transcriptionally tuned to accommodate the functions of associated neurons. Our genetic ablation studies in mice revealed that satellite glia play a key role in modulating the activity of sympathetic neurons (Mapps et al., 2022a). Mice lacking these glia exhibit enhanced sympathetic neurotransmission and perturbed autonomic responses such as elevated heart rate. This unexpected finding brings a new perspective to the basis of sympathetic hyperactivity in disorders such as cardiovascular disease and hypertension.
Current studies: Through single cell RNA-seq, we identified 4 distinct satellite glial populations in sympathetic ganglia and defined unique gene signatures for each population (Mapps et al., 2022b). We are thus poised to target individual satellite glial cell sub-types genetically for imaging and ablation studies. We are addressing how specific sub-types are specified during development, how they become intimately associated with their neuronal neighbors, and how glia-neuron interactions influence the formation, function, and maintenance of sympathetic circuits.
Credit: Aurelia Mapps
Credit: Joselyn Ramírez-Guiterrez
Mapps et al., 2022a
Regulated transcytosis of neurotrophin receptorsReduced axon innervation of sympathetic targets in TrkAR685A miceConnor et al., 2023